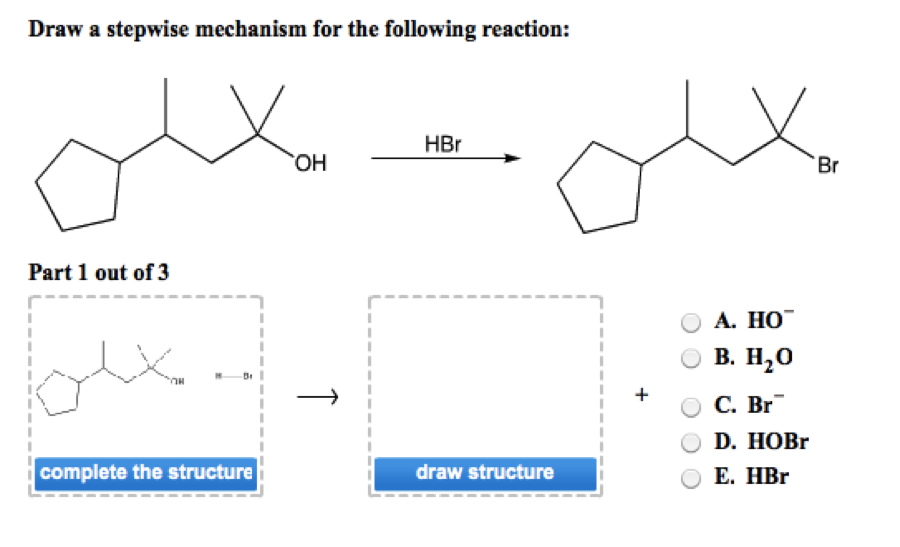
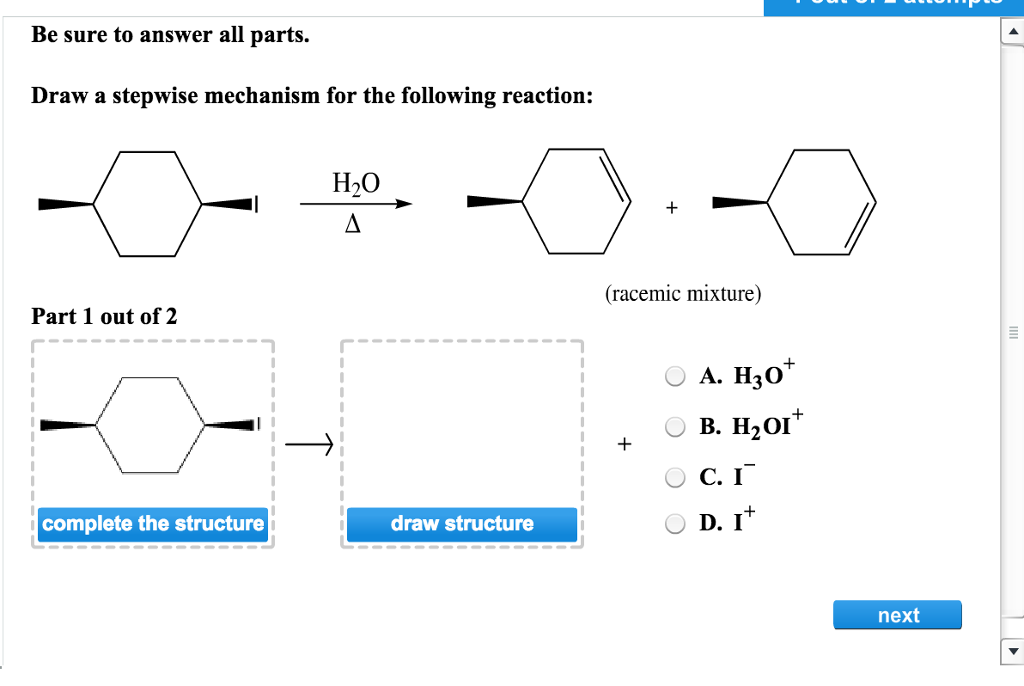
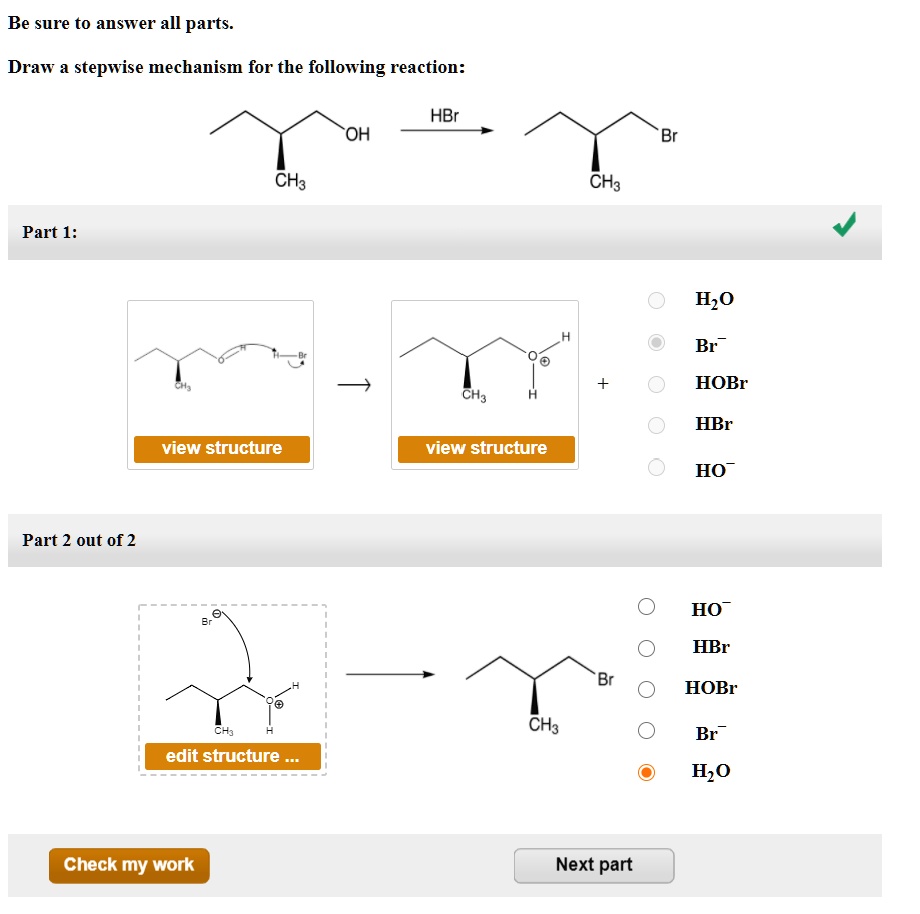
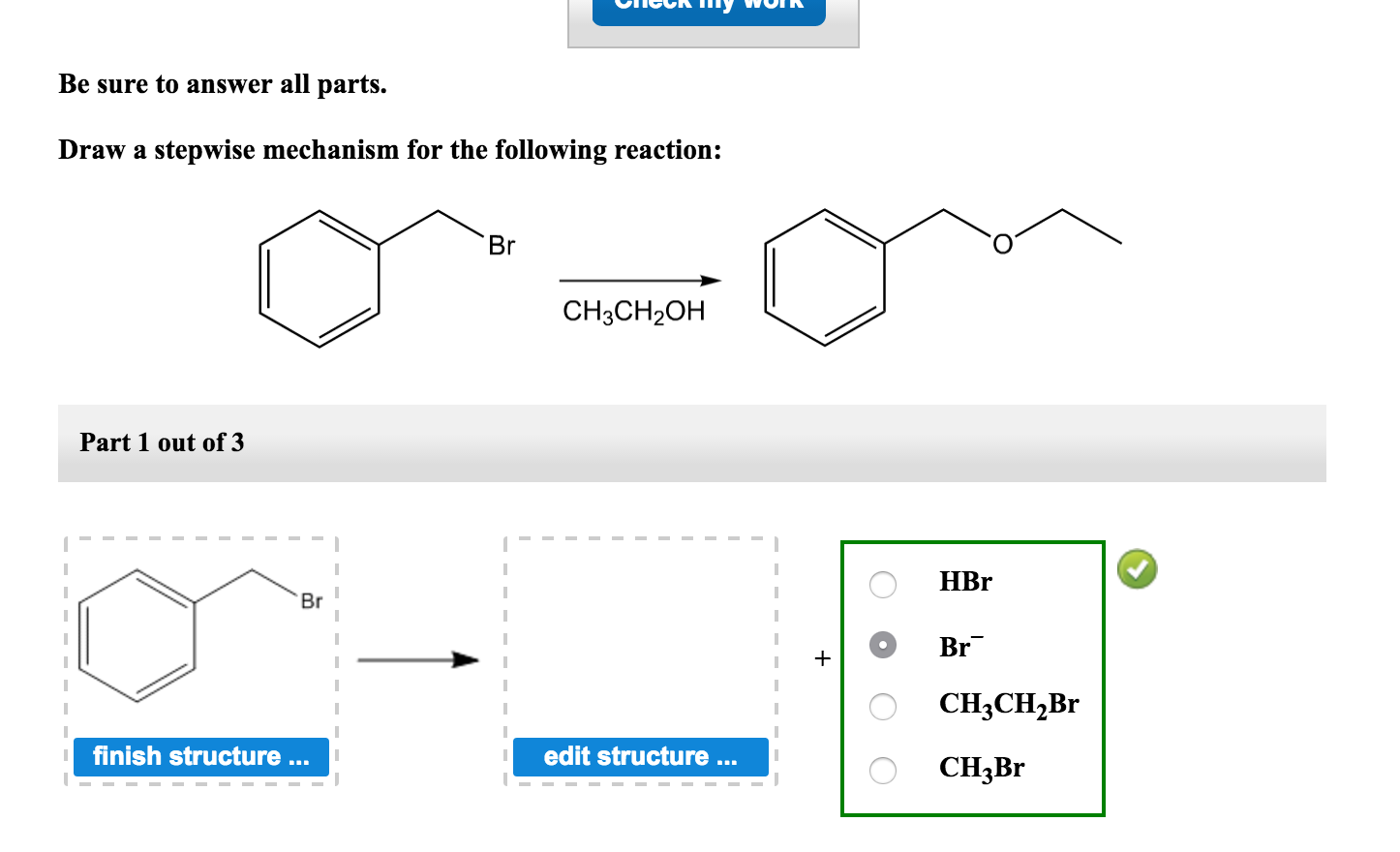
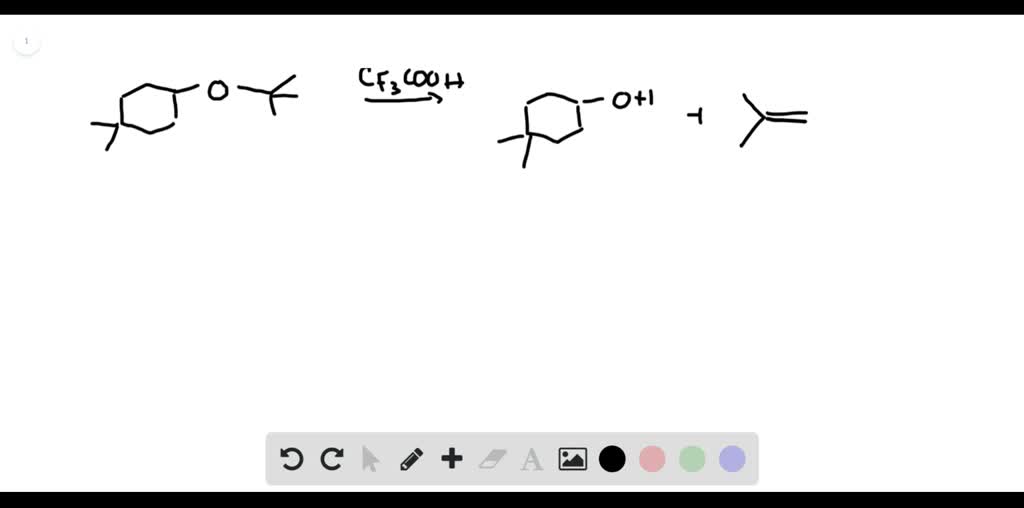
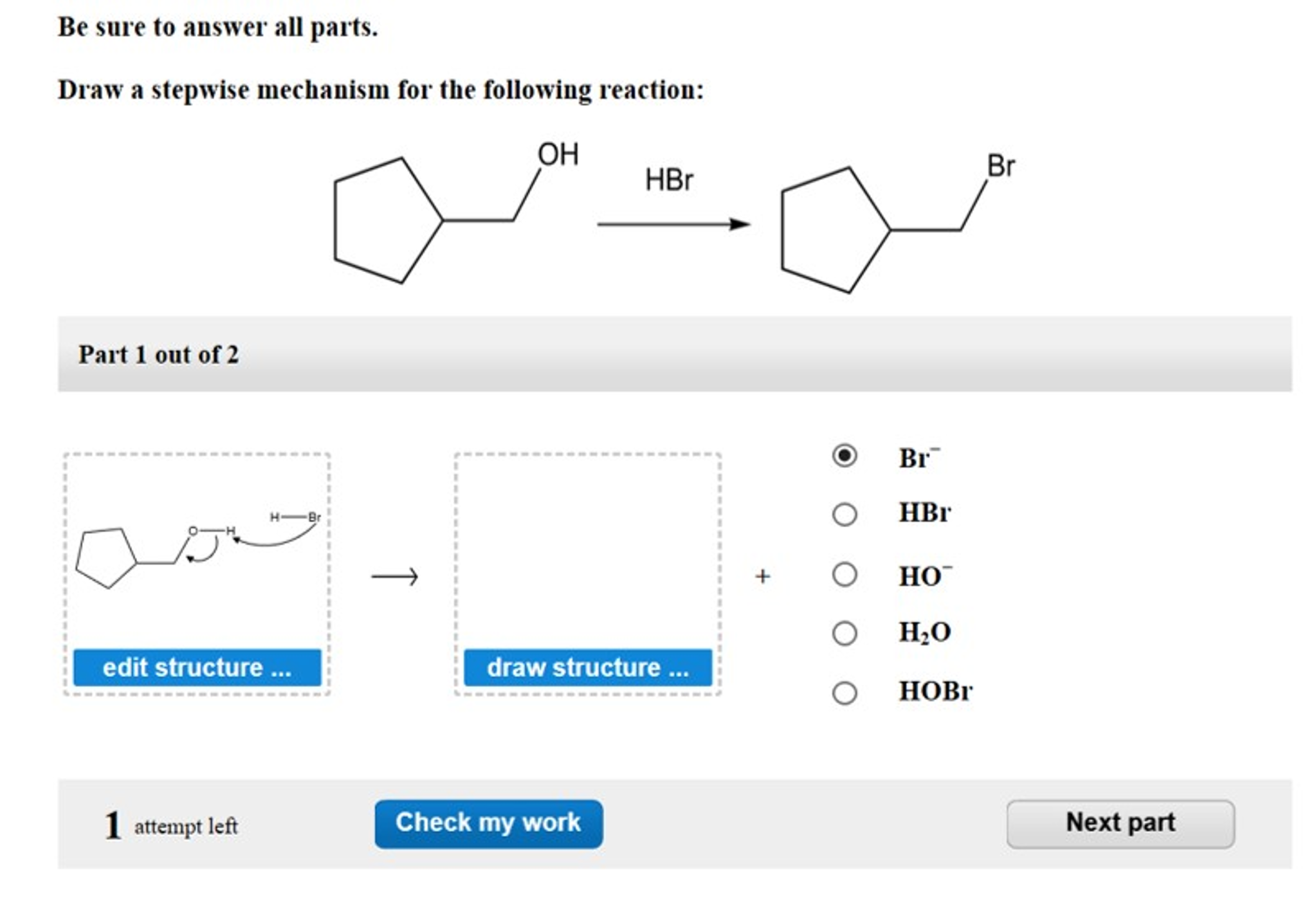
Draw A Stepwise Mechanism For The Following Reaction
Draw A Stepwise Mechanism For The Following Reaction - Web the first step for the given reaction involves the addition of halogen to the acetylene molecule and results in the formation of a cyclic intermediate. 100% (8 ratings) share share. Be sure to answer all parts. A reaction that occurs in two or more elementary steps is called a multistep or complex reaction. The decomposition of ozone, for example, appears to follow a mechanism with two steps: Derive the rate law consistent with a given reaction mechanism. View the full answer step 2. Draw a stepwise mechanism for the following reaction: This is a markovnikov's addit. Ch,ch,br hbr br view structure view structure ch, br part 2: Draw a stepwise mechanism for the following reaction: Br h view structure part 2 out of 2 br edit structure. Here’s the best way to solve it. This is a markovnikov's addit. There are 2 steps to solve this one. Web a balanced equation indicates what is reacting and what is produced, but it reveals no details about how the reaction actually takes place. Web this reaction combines two processes together: In an internal combustion engine, for example, isooctane reacts with oxygen to give carbon dioxide and water: Oh soci, pyridine + so2 + hci. Web the first step for the given reaction involves the addition of halogen to the acetylene molecule and results in the formation of a cyclic intermediate. Draw a stepwise mechanism for the following reaction: Web the first step for the given reaction involves the addition of halogen to the acetylene molecule and results in the formation of a cyclic intermediate. Web the reaction mechanism (or reaction path) is the process, or pathway, by which a reaction occurs. View structure view structure part 3 out of 3. In an internal combustion engine, for example, isooctane reacts with oxygen to give carbon dioxide and water: A reaction mechanism is the sequence of elementary steps by which a chemical reaction occurs. Draw a stepwise mechanism for the following reaction: A reaction that occurs in two or more elementary steps is called a multistep or complex reaction. Web draw a. In an internal combustion engine, for example, isooctane reacts with oxygen to give carbon dioxide and water: Draw a stepwise mechanism for the following reaction: Be sure to answer all parts. Web this reaction combines two processes together: I need help on part two. Draw a stepwise mechanism for the following reaction: A reaction that occurs in two or more elementary steps is called a multistep or complex reaction. Just need help on the last one. Here first oxygen attack on the socl2 and chloride ion r. There are 2 steps to solve this one. Here first oxygen attack on the socl2 and chloride ion r. In an internal combustion engine, for example, isooctane reacts with oxygen to give carbon dioxide and water: Web draw a stepwise mechanism for the following reaction: Web the reaction mechanism (or reaction path) is the process, or pathway, by which a reaction occurs. Oh soci, pyridine + so2 +. Here’s the best way to solve it. View structure view structure part 3 out of 3 edit structure. Here first oxygen attack on the socl2 and chloride ion r. View the full answer step 2. 100% (8 ratings) share share. This is a markovnikov's addit. Just need help on the last one. Oh soci, pyridine + so2 + hci. View the full answer step 2. Web this reaction combines two processes together: A reaction intermediate is a chemical species that is formed in one elementary step and consumed in a subsequent step. A reaction mechanism is the sequence of elementary steps by which a chemical reaction occurs. Web a chemical reaction often occurs in steps, although it may not always be obvious to an observer. O 3(g) o 2(g) + oo +. 100% (8 ratings) share share. Here’s the best way to solve it. Here’s the best way to solve it. Draw a stepwise mechanism for the following reaction: Just need help on the last one. Identify the nucleophilic site on the alcohol and the electrophilic site on thionyl chloride (socl2). There are 3 steps to solve this one. Web write a balanced chemical equation for a process given its reaction mechanism. There are 2 steps to solve this one. I need help on part two. Web draw a stepwise mechanism for the following reaction: Be sure to answer all parts. Be sure to answer all parts. Derive the rate law consistent with a given reaction mechanism. Web the reaction mechanism (or reaction path) is the process, or pathway, by which a reaction occurs. Web draw a stepwise mechanism for the following reaction: Just need help on the last one. There are 2 steps to solve this one. Begin the mechanism by protonating the epoxide ring.) solution. Draw a stepwise mechanism for the following reaction: The water molecule then acts as a nucleophile and leads to the formation of carbocation intermediate, which is. There are 2 steps to solve this one. O 3(g) o 2(g) + oo + o 3(g) 2o 2(g) we call each step in a reaction mechanism an elementary reaction. A reaction intermediate is a chemical species that is formed in one elementary step and consumed in a subsequent step. There are 3 steps to solve this one. Web write a balanced chemical equation for a process given its reaction mechanism.Solved Draw a stepwise mechanism for the following reaction
Solved Draw A Stepwise Mechanism For The Following Reacti...
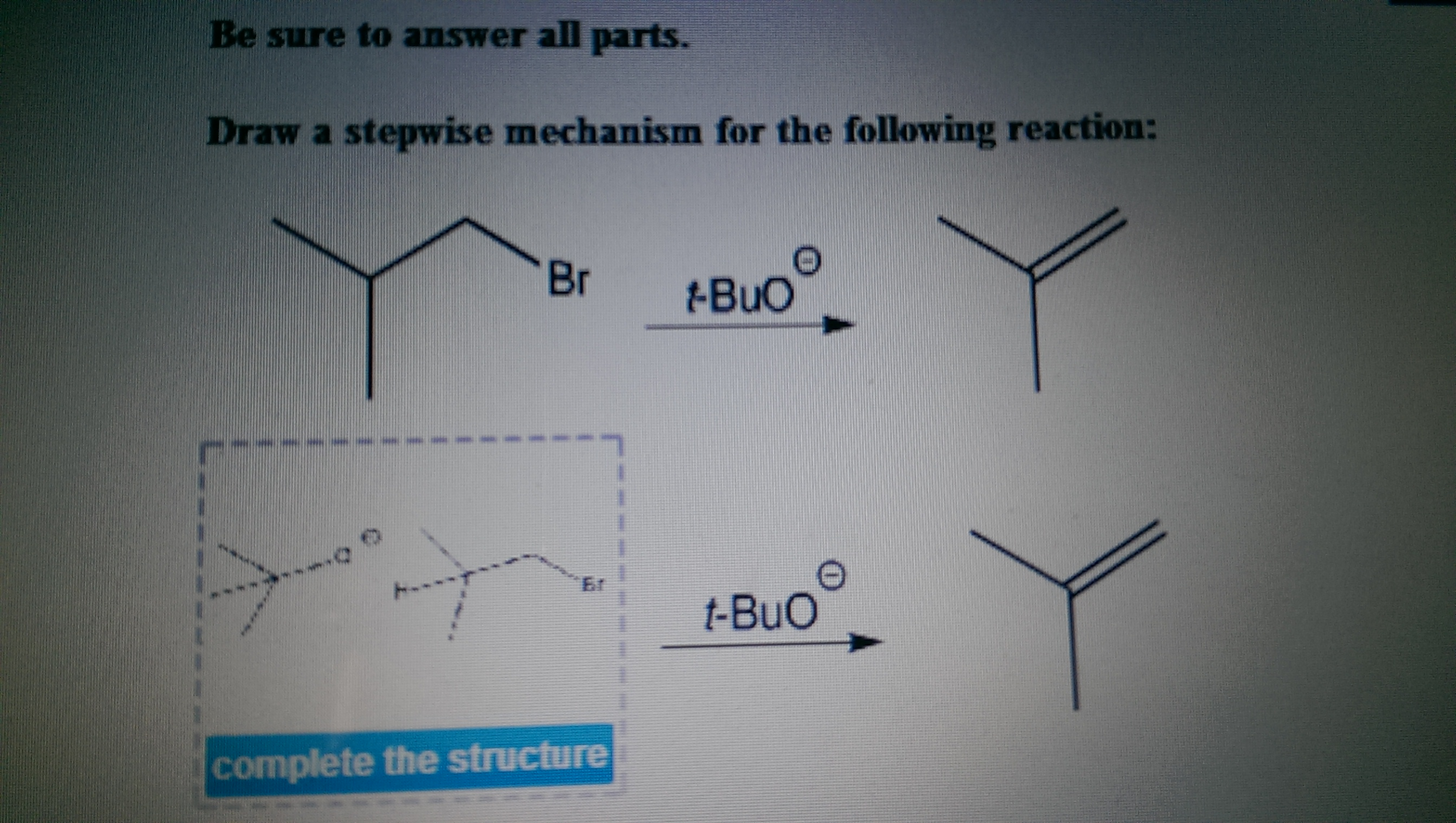
Draw A Stepwise Mechanism For The Following Reaction Hbr Sexiz Pix
Solved Draw a stepwise mechanism for the following reaction
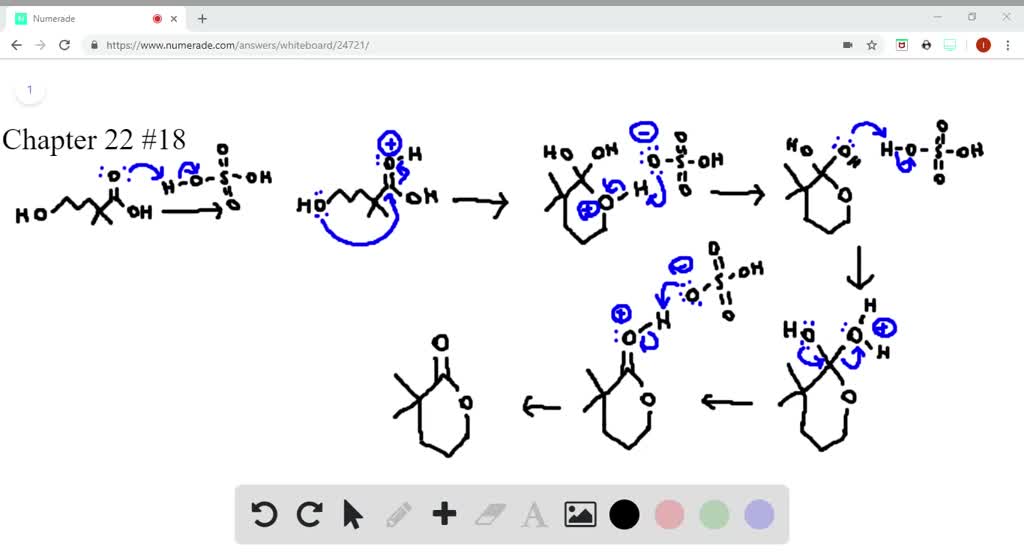
SOLVEDDraw a stepwise mechanism for the following reaction.
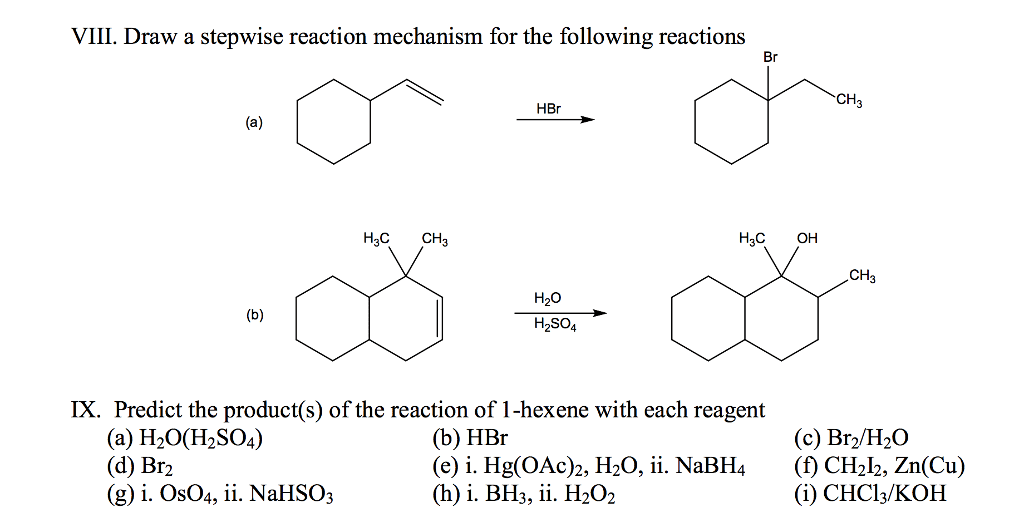
OneClass VIII. Draw a stepwise reaction mechanism for the following
Draw The Mechanism For The Following Reaction
Solved Draw a stepwise mechanism for the following reaction
SOLVEDDraw a stepwise, detailed mechanism for the following reaction.
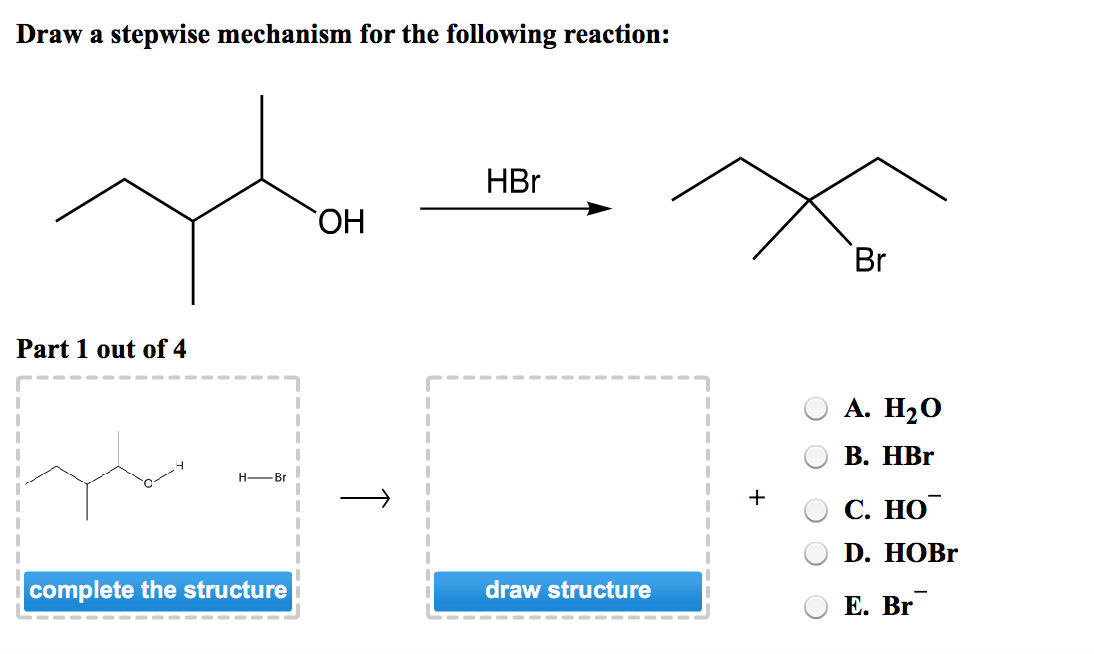
Draw a stepwise mechanism for the following reaction
I Need Help On Part Two.
A Mechanism Refers To The Series Of Steps That The Reagents Undergo During A Chemical Reaction.
Web Draw A Stepwise Mechanism For The Following Reaction:
Here First Oxygen Attack On The Socl2 And Chloride Ion R.
Related Post: