Draw The Electron Configuration For A Neutral Atom Of Cobalt
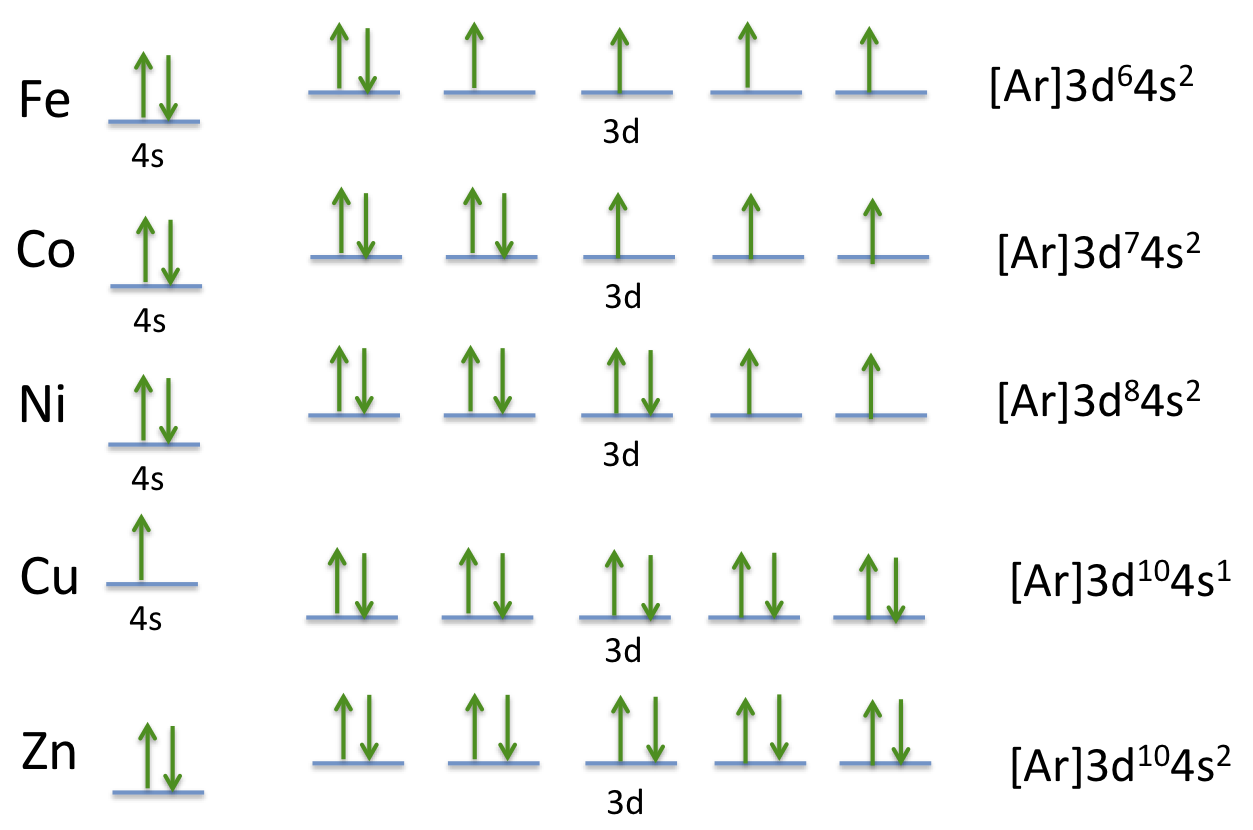
Draw The Electron Configuration For A Neutral Atom Of Cobalt - Web to write the configuration for the cobalt ions, first we need to write the electron configuration for just cobalt (co). The cobalt (iii) ion, however, has lost 3. In this article, we will study how are electrons arranged in different shells and subshells in an atom of cobalt. Energy х this problem has been solved! 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2. By knowing the electron configuration of an element, we can predict and explain a great deal of its chemistry. Determine the number of electrons in a neutral cobalt atom. The last three have only 1 electron. Web give the electron configuration for a neutral atom of cobalt (co) using an orbital diagram (the kind with lines or boxes for orbitals and arrows to show the electrons). Then writing the configuration of the remaining electrons: Web the electron configuration of an element is the arrangement of its electrons in its atomic orbitals. We first need to find the number of electrons for the co atom. 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁷ 4p¹. Draw the electron configuration for a neutral atom of cobalt. In terms of energy, as you ascend from the bottom to the top of the electron configuration diagram, the energy increases. Web the electron configuration of a neutral cobalt atom is: You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The electron configuration of a neutral cobalt atom is [ar] 4s2 3d7.step 2/42. Justify the anomalies of the electron configurations in transition metals using magnetism experimental data. Web electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of sulfur. Here’s the best way to solve it. We first need to find the number of electrons for the co atom. Web electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of sulfur. Draw the electron configuration for a neutral atom of cobalt. In terms of energy, as you. Determine the atomic number of cobalt from the periodic table. Web determine the electron configuration of ions. Then writing the configuration of the remaining electrons: Web electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of sulfur. In this video we'll use the periodic table to help us. Web two of the three d orbitals have two electrons. 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2. This represents the distribution of electrons in the atomic orbitals from lowest to highest energy levels. So, the more accurate electron configuration is: Web to write the configuration for the cobalt ions, first we need to. The electron configuration of a neutral cobalt atom is [ar] 4s2 3d7.step 2/42. 1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁸ 4s¹. 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁷ 4p¹. Web the electron configuration of a neutral cobalt atom is: Energy х this problem has been solved! This represents the distribution of electrons in the atomic orbitals from lowest to highest energy levels. Web the electron configuration of a neutral cobalt atom is: Web electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of sulfur. 1s 2 2s 2 2p 6 3s 2 3p 6. Here’s the best way to solve it. Web electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of sulfur. Web the electron configuration for a neutral cobalt atom, represented using the aufbau principle, is 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁷. By knowing the electron configuration of an. Find the atomic number of cobalt from the periodic table. So, the more accurate electron configuration is: Web the electron configuration of a neutral atom of cobalt is 1s2 2s2 2p6 3s2 3p6 4s2 3d7. By knowing the electron configuration of an element, we can predict and explain a great deal of its chemistry. Since the atomic number is 27,. This electron configuration allows the 3 single electrons to all spin the same direction creating an electrical current which causes cobalt to be one of the naturally magnetic elements. In terms of energy, as you ascend from the bottom to the top of the electron configuration diagram, the energy increases. Web draw the electron configuration for a neutral atom of. Web the electron configuration of a neutral cobalt atom is: Determine the atomic number of cobalt from the periodic table. Web for cobalt it's argon (which has filled electron shells with maximum number of electrons); Find the atomic number of cobalt from the periodic table. Web determine the electron configuration of ions. Web the electron configuration of cobalt is: Then writing the configuration of the remaining electrons: Web the electron configuration of an element is the arrangement of its electrons in its atomic orbitals. Web determine the electron configuration of ions. Check the atomic number (29) and atomic mass (63.546) of copper. [ a r ] 3 d 7 4 s 2 \mathrm{[ar]3d^74s^2} [ ar ] 3 d 7 4 s 2 Web for cobalt it's argon (which has filled electron shells with maximum number of electrons); Web the electron configuration of a neutral atom of cobalt is 1s2 2s2 2p6 3s2 3p6 4s2 3d7. 1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁸ 4s¹. Determine the atomic number of cobalt from the periodic table. Web the electron configuration for a neutral cobalt atom, represented using the aufbau principle, is 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁷. The cobalt (iii) ion, however, has lost 3. Then writing the configuration of the remaining electrons: Web electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of sulfur. Web the electron configuration of an element is the arrangement of its electrons in its atomic orbitals. Web draw the electron configuration for a neutral atom of cobalt. This electron configuration allows the 3 single electrons to all spin the same direction creating an electrical current which causes cobalt to be one of the naturally magnetic elements. The atomic number of cobalt is 27, which means a neutral cobalt atom has 27 electrons. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Since the atomic number is 27, there are 27 electrons in a neutral cobalt atom. Check the atomic number (29) and atomic mass (63.546) of copper.Electron Configuration Of Cobalt
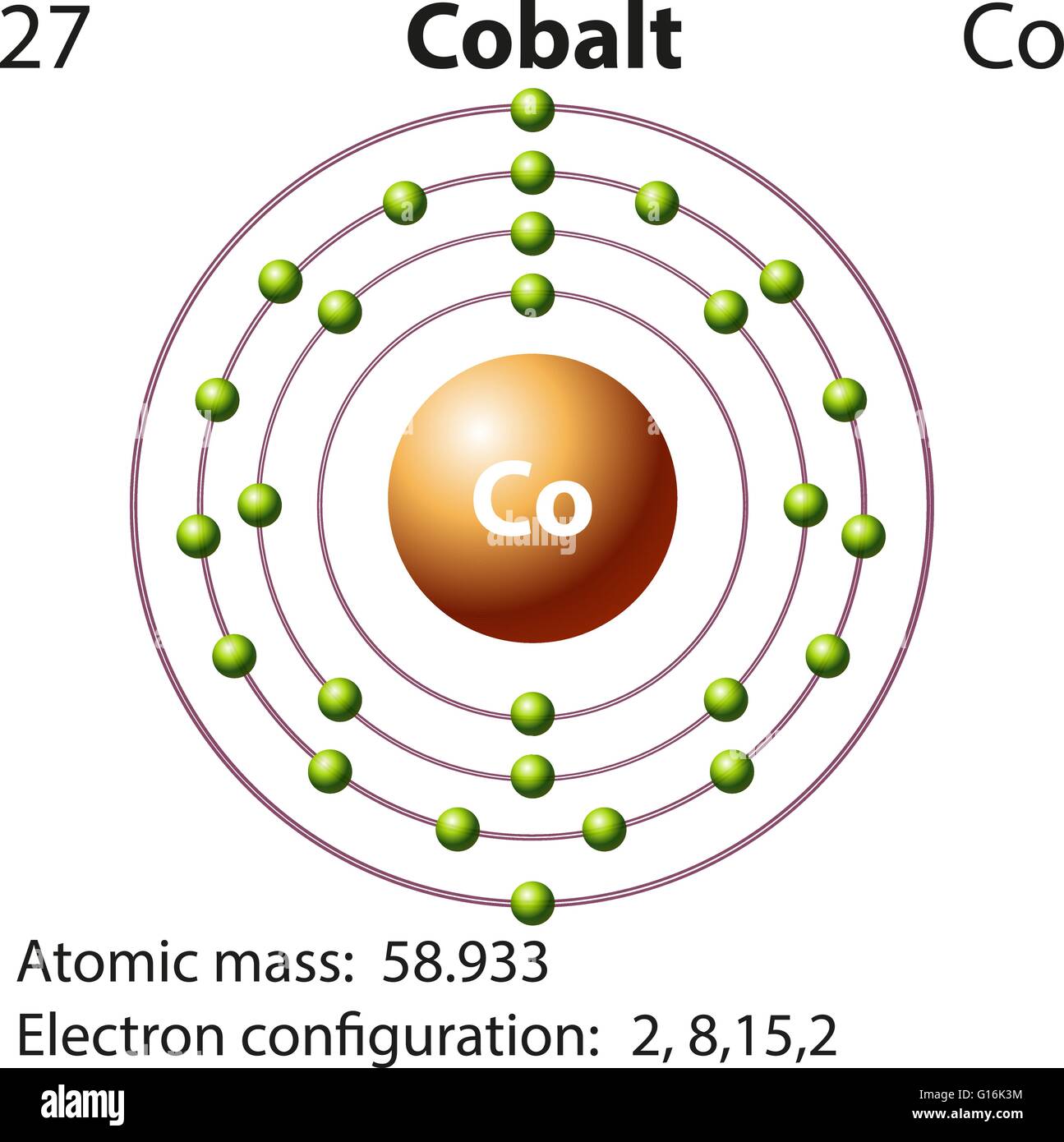
Cobalt Electron Configuration (Co) with Orbital Diagram
Draw The Electron Configuration For A Neutral Atom Of Cobalt biketrip
SOLVED Draw the electron configuration for a neutral atom of cobalt
Solved Draw the electron configuration for a neutral atom of
What is the electron configuration for a neutral atom of cob Quizlet
(Get Answer) Draw The Electron Configuration For A Neutral Atom Of
SOLVED Draw the electron configuration for a neutral atom of cobalt
Symbol and electron diagram for Cobalt illustration Stock Vector Image
Cobalt Electron Configuration (Co) with Orbital Diagram
Web The Electron Configuration Of Cobalt Is:
Write The Electron Configuration For A Neutral Atom Of Manganese.
In This Video We'll Use The Periodic Table To Help Us Write The Notation For.
We First Need To Find The Number Of Electrons For The Co Atom.
Related Post: